Responsible for 17.9 million of deaths in 2015 (32% of total deaths worldwide), heart disease is a major killer. The improvement of available treatments is essential if we are to reduce its impact. Prof. Michael Geeves, from the School of Biosciences, is one of those scientists working to try and achieve this. He has recently been awarded an EU grant for a new project involving the design of a physiological model of the human heart in collaboration with 10 other institutions across Europe and the USA. Prof Geeves gives us an insight as to what the project is really about, and why it is so important.
Why did Professor Geeves decide to go into research?
Research consists of always testing new ideas. Sometimes they work and sometimes they don’t – but both are quite exciting, as even when you get a ‘’wrong’’ answer, you have to investigate to realize what went wrong. I think it’s the discovery process that I enjoy the most.
What are the discoveries that have led him to this project?
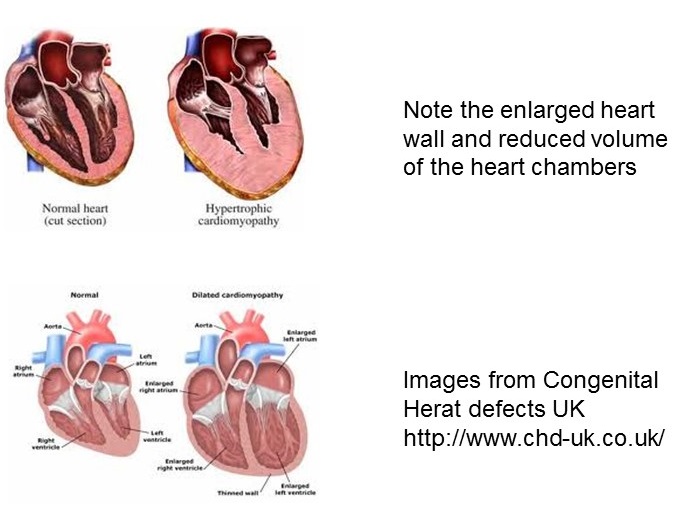
It’s been developing over a long period of time. My work largely revolves around the proteins involved in muscle contraction in the heart, that is, actin and myosin. I have been studying these processes for about 40 years. We are trying to put these together to build a model of how the heart muscles contract. This is very slow work as the heart is quite a complex system.
10 different institutions are involved in this new grant. There are mathematicians, computer scientists, biochemists and, clinicians… We have been working to build a model of how a single heat cell contracts – this is hard enough, but the complexities of building an entire heart model are much larger. Unlike skeletal muscle, the heart goes through a complex process of contraction with different phases of contraction across the heart during each heart-beat.
This is going to be a long-term project. The hope is that some of the elements we put together along the way become useful in themselves, but the complete thing could take 10 years or more.
How would he explain the importance of this project to someone without a science background?
Heart disease is the biggest killer of people – particularly young people under 30. Most people who die young die from accidents rather than from disease, with the exception of heart disease. Trying to find ways of preventing these deaths is quite important, and the development of drugs to treat cardiovascular disease is key. This new physiological model of the heart will hopefully aid the drug development process.
Aside from its potential uses in the pharmaceutical industry, does the project have other applications?
Absolutely. Aside from assisting with the understanding of drug targets to treat cardiovascular disease, it also has a diagnostic application. Clinicians would be able to use it to better diagnose heart conditions and choose an appropriate treatment for the patient.
New paper in press Journal of Biological Chemistry
Dilated cardiomyopathy myosin mutants have reduced force-generating capacity.
Zoltan Ujfalusi1,7,#, Carlos D. Vera2,#, Srbolujub M. Mijailovich3, Marina Svicevic4, Elizabeth Choe Yu5, Masataka Kawana6, Kathleen Ruppel6, James A. Spudich6, Michael A. Geeves1,* & Leslie A. Leinwand2,*
A collaboration between 1. UKC, 2. University of Colorado USA , 5 & 6. Stanford University USA, 3. Illinois Institute of Technology USA, 4. University of Kragujevac, Serbia, 7. University of Pecs, Hungary.
*Corresponding authors